Structure of a hantavirus protein as a promising model for drug design
X-ray crystallography provides drug template against disease transmitted by small rodents
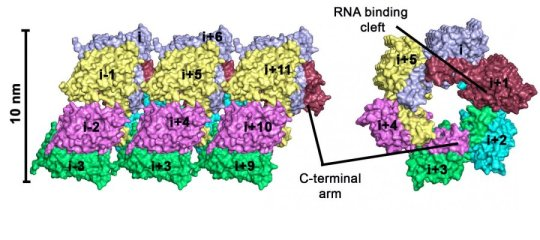
Left: Hexameric rings form a tube of viral capsid. Right: view from the other side of the protein oligomer.
Credit: Daniel Olal/MDC
Bank voles are small rodents that are not dangerous by themselves, but their excreta can contain one of the dangerous hantaviruses. While bank voles are unaffected by the infection, hantaviruses can cause potentially fatal diseases in humans for which no treatments exist. In central and northern Europe, infection is accompanied by fever, headache, or even renal failure. The strain that occurs in East Asia — the Hantaan virus — is even more dangerous: up to five percent of infected patients die of hemorrhagic fever, renal failure, or severe respiratory disorders.
Dr. Daniel Olal and Prof. Oliver Daumke of the MDC in Berlin have now analyzed the nucleoprotein of the Hantaan virus by means of X-ray crystallography and identified its three-dimensional structure. Olal and Daumke have worked out how individual nucleoproteins oligomerize in the presence of RNA molecules, and they have found hexameric circular complexes. “We already know about cellular defense mechanisms that inhibit viral growth. We think that the circular structures we have identified could play a part in this,” says Olal.
The nucleoprotein plays an important part in replication of the viral genome. If its function is disrupted, the cell cannot produce functional virus particles. This protein is therefore an ideal target structure for future drugs. “Our structure could be useful for the design of small molecules that specifically block the nucleoprotein,” says Olal. The researchers have identified three binding pockets on the surface of the protein that could serve as docking sites for such compounds. Olal is positive that such drugs will be developed in the future: “After all, the technology for finding candidates for drug development is getting better all the time.”
0 Comments