Gene therapy pioneer James Wilson uses CRISPR/Cas9 to target liver disease
One of the pioneers in the whole gene therapy movement of the past 35 years has combined his knowledge of viral vectors with the hot new CRISPR/Cas9 tech to tackle a rare genetic liver disease. And his work with rodents highlighted both the promise of this new technology as well as an unexpected hurdle.
James Wilson at the University of Pennsylvania is virtually the father of adeno-associated virus–or AAV–vectors used to deliver gene therapies. After years of up and down progress, including an early patient death that slowed research in gene therapies for decades, his technology was in-licensed by ReGenX, which has since outlicensed it to a group of biotechs pursuing new gene therapy programs.
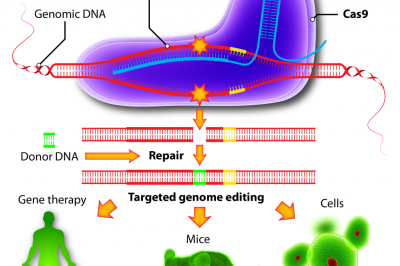
In what is described as a first in research circles, he used an AAV vector to deliver the components of CRISPR/Cas9 gene editing tools into a mouse model of a rare metabolic urea-cycle disorder, triggered by a deficiency in the ornithine transcarbamylase (OTC) enzyme. When one of a series of enzymes is missing or “deficient,” ammonia can accumulate in the blood and circulate into the brain, where it can badly damage brains. OTC deficiency occurs in one in every 40,000 births.
Wilson’s group used one AAV to deliver a Cas9 enzyme–the cutting tool–specifically into liver cells. Another vector carried a guide RNA to the specified spot and donor DNA to correct the mutation in a cut-and-paste approach that is spreading around academic labs like wild fire.
In a research article published in Nature Biotechnology, Wilson says his team achieved a 10% reversion in the liver cells of newborn mice, with an increased survival rate for challenged rodents and a high death rate in the untreated group of newly born mice. In adult mice, though, the team say that the experiment didn’t work as planned, highlighting a needed correction in the process. And now they’re following up with new approaches to see how that might work.
“Correcting a disease-causing mutation following birth in this animal model brings us one step closer to realizing the potential of personalized medicine,” said Wilson, a professor of medicine and director of the Orphan Disease Center at Penn. “Nevertheless, my 35-year career in gene therapy has taught me how difficult translating mouse studies to successful human treatments can be. From this study, we are now adjusting the gene-editing system in the next phases of our investigation to address the unforeseen complications seen in adult animals.”
0 Comments